PH CalculationsPage
1
1
Slide 1
pH Calculations
Soren Sorensen
Slide 2
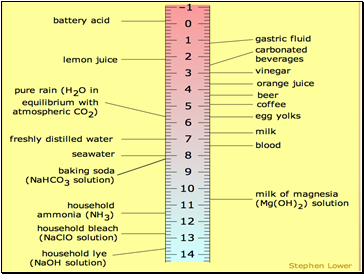
pH Scale
Slide 3
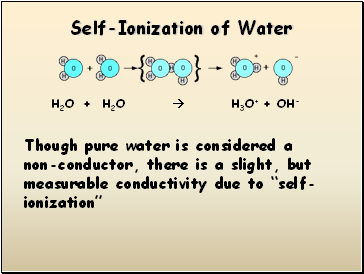
Self-Ionization of Water
H2O + H2O H3O+ + OH-
Though pure water is considered a non-conductor, there is a slight, but measurable conductivity due to “self-ionization”
Slide 4
Kw – Ionization Constant for Water
In pure water at 25 C:
[H3O+] = 1 x 10-7 mol/L
[OH-] = 1 x 10-7 mol/L
Kw is a constant at 25 C:
Kw = [H3O+][OH-]
Kw = (1 x 10-7)(1 x 10-7) = 1 x 10-14
Slide 5
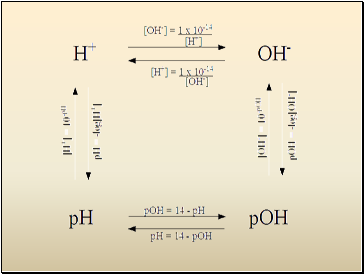
Calculating pH, pOH
pH = -log10(H3O+)
pOH = -log10(OH-)
Relationship between pH and pOH
pH + pOH = 14
Finding [H3O+], [OH-] from pH, pOH
[H3O+] = 10-pH
[OH-] = 10-pOH
Slide 6
pH + pOH = 14
Slide 7
Contents
Last added presentations
- Newton’s laws of motion
- Radioactivity and Nuclear Reactions
- Newton’s third law of motion
- Ch 9 Nuclear Radiation
- Radiation Safety and Operations
- Thermal Energy
- The Effects of Radiation on Living Things
© 2010-2025 powerpoint presentations